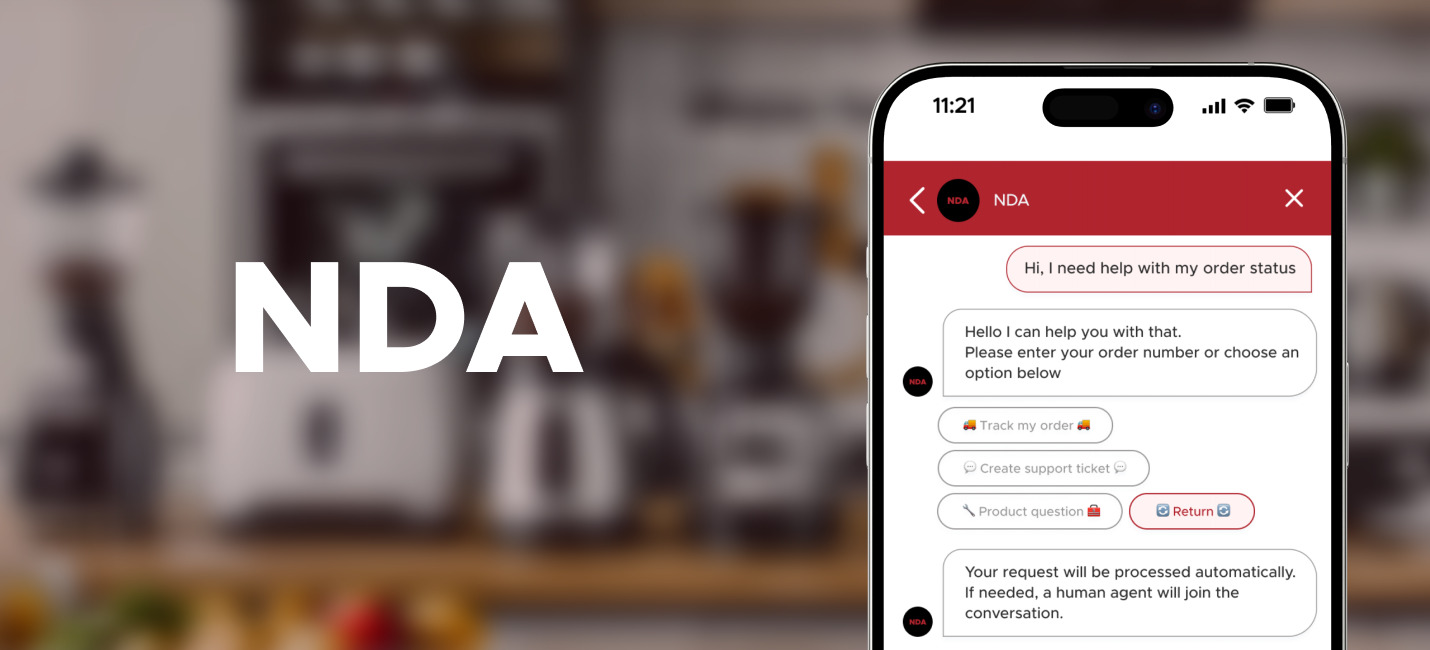
Chats with the chatbot
NEVA. Genetic Testing Chatbot for Natera Inc.

Natera is a global leader in cell-free DNA (cfDNA) testing that has been offering genetic testing for more than 10 years. With over 1300 employees in 90 countries, it provides proprietary testing services to physicians, researchers, and clinicians in women's health, transplant and cancer, and genetic laboratories through its cloud-based software platform.
There are now more than 75,000 genetic tests. That comes to over 10 new tests that enter the market every day. According to Global Market Insights, the global genetic testing market is set to exceed $22 billion by the end of 2024.
The price of these tests has significantly decreased, now varying from less than $100 to over $2,000, depending on the test's type and complexity.
Therefore, patients are increasingly opting for genetic tests that evaluate the risk of diseases like breast cancer, Alzheimer's, Parkinson's, diabetes, and other conditions, particularly when there is a family history involved. Prenatal genetic testing has also surged in popularity, particularly for screening conditions like Down syndrome, cystic fibrosis, and other chromosomal abnormalities.
However, too few genetics professionals exist to meet the increased demand for genetic information. This is why our client contacted us — to help them address their patient growth by providing high-quality genetic information to more patients in a scalable way.
Our Contribution to Success
We developed a healthcare chatbot that assists the company in delivering genetic education. The chatbot guides and educates patients about genetic testing and helps patients get reliable information faster and more conveniently. Also, the chatbot sends detailed explanations of test results through the patient's portal and a patient can book a meeting with a genetic counselor.
Meet NEVA. You can read how to work with NEVA here.
Key Results
Currently, there are more than 4000 chats with the bot, and the success rate is over 80%, which means that most of the patients who start the interaction complete the whole chatbot flow.
These results show that most of the patients self-assisted their needs for the test results explanation which saved valuable counselor time. Given these results, Natera can help providers meet the demand for genetic information, and expand remote access to genetics services in a scalable way.
Completion rate — users who started and finished the entire conversation journey
The Winning Formula: How Did We Achieve Success
Planning
Together with Natera, we discussed their needs, goals, and challenges to outline the work scope, create a roadmap with the milestones, and approve the estimate. We decided to start small and build a Proof of Concept (POC) to validate the chatbot idea, and after client’s approval, we began scaling it into a full version of the chatbot.
Development
We started by outlining the initial chatbot flow and conversational design based on the documentation that the company provided. When the first version of the flow was approved with the client, we moved on to the actual coding. Here are technologies we had used:
Functionalities
The company's current patients can log in to the portal and chat with the bot to discuss specific genetic test results.

The pre- and post-test processes guide patients through the specifics of each test, offering detailed information about the test itself, the procedure, and who stands to benefit from it. The user can create a patient account in the clinic or get a personalized link to talk with the chatbot to learn more about a particular genetic test or undergo a test to uncover initial susceptibility to genetic diseases.
The information is gathered and can be forwarded straight to the user's family doctor, who will then refer the individual to a specialist for a more in-depth explanation and medical evaluation. Post-test functionality is designed for the company's patients to enable round-the-clock access to information and to help the counselors save time explaining the initial results and invest more in complicated tasks and test results.
“The team is communicative, flexible, and skilled. BotsCrew is always easy to work with and delivers user-friendly solutions.”
Now, together with the client's team, we are working on the next versions of the chatbot to increase patient engagement, develop new functionalities and expand the suite of products.
Transform AI from a Risk into a Strategic Advantage.
Partner with BotsCrew to build AI that is fair, transparent, and aligned with your company's values.
Contact usThe tests described have been developed and their performance characteristics determined by the CLIA-certified laboratory performing the test. The tests have not been cleared or approved by the US Food and Drug Administration (FDA). Although FDA is exercising enforcement discretion of premarket review and other FDA legal requirements for laboratory-developed tests in the US, certification of the laboratory is required under CLIA to ensure the quality and validity of the tests. CAP-accredited, ISO 13485 certified, and CLIA certified. © 2020 Natera, Inc. All Rights Reserved.